Oxygen (O2)
Oxygen is vital to life on our planet. It’s also essential for a wide range of industries that support daily life, including steel manufacturing, chemicals, paper manufacturing, and medicine. However, it also poses certain hazards. Here we’ll present some potentially life-saving facts about oxygen.
Composition of air
Accounting for approximately 21 % of Earth’s atmosphere, oxygen is essential for humans and other creatures.
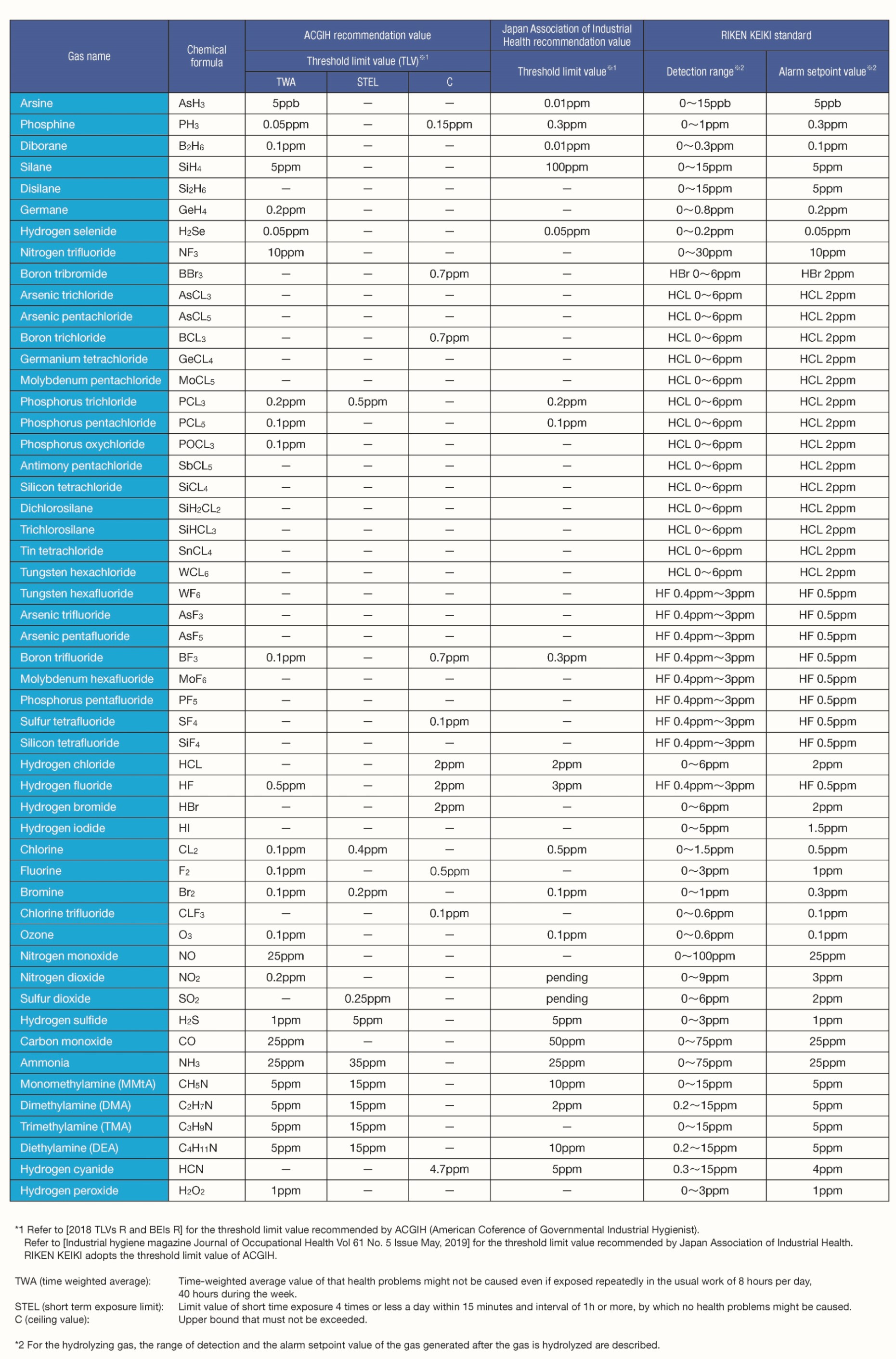
| Atmospheric gas component | Percentage by volume | Partial pressure |
|---|---|---|
| Oxygen | 20.93 % | 151.1 mmHg |
| Nitrogen | 78.09 % | 593.5 mmHg |
| Carbon dioxide | 0.03 % | 0.2 mmHg |
| Argon and other rare gases | 0.95 % | 7.2 mmHg |
| Total | 100.00 % | 760.0 mmHg |
Definition of anoxia (oxygen deficiency)
Anoxia is the condition in which the concentration of oxygen in the air is below 18 %.
As defined in Article 2 of the Ordinance on Prevention of Anoxia, etc. (in Japan)

Data on industrial accidents involving oxygen deficiency (1990 - 2020) (in Japan)
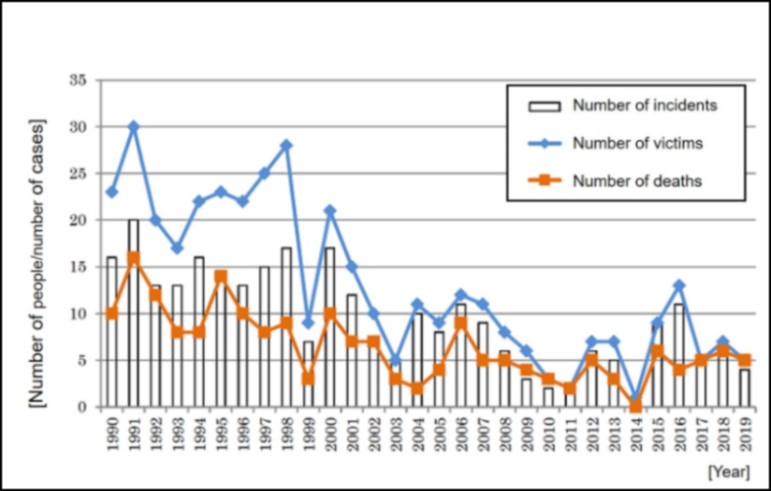
Extract from Ministry of Health, Labour and Welfare Basic Industrial Safety Notification No. 0707-3
- Main causes of oxygen deficiency -
Oxygen consumption
Oxidation of metals, ore, and subsoil iron, human respiration, respiration by cereals, fruits, vegetables, and timber, oxidation of paints and solvents
Influx of inert gases or coolants
Influx of N2 gas (including liquid nitrogen), argon gas, or CO2, influx or buildup of coolants such as fluorocarbons
Release of combustible gases
Release of methane gas during excavation, gas leaks from fuel gas pipes
Excessive oxygen
Higher oxygen concentrations will increase the size and temperature of flames, lower the ignition temperature of combustible materials, and increase combustion speed. All this results in an increased risk of fire.
For humans, continuous inhalation of high oxygen concentrations can lead to convulsions, loss of consciousness, and, in the worst case, death.