Work function and ionization potential measurements
Here we describe how work function and ionization potential are measured.
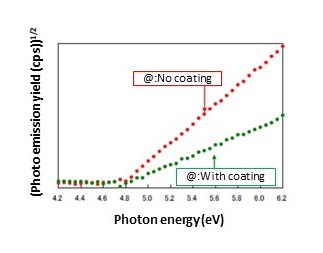
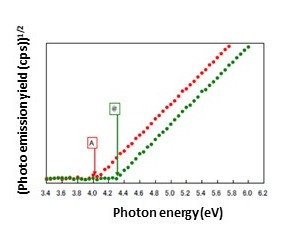
When a test sample surface is irradiated with UV light, the emitted photoelectron count varies depending on the energy of the UV light.
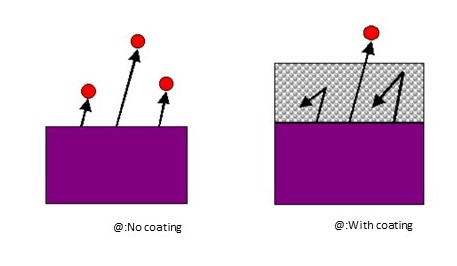
As in A, no photoelectrons are emitted (Fig. 2) when the UV energy is low (Fig. 1).
However, as in B, if the UV energy becomes sufficiently high, valence band electrons are excited to higher energy levels than the vacuum level (Fig. 1), resulting in photoemission (Fig. 2).
The threshold energy for photoemission (@ in Fig. 2) marks the difference between vacuum level and valence band maximum energy (@ in Fig. 1). In metals and other cases in which the valence band maximum energy and Fermi level are the same, the photoemission threshold energy corresponds to the work function.
In semiconductors and other cases in which the valence band maximum energy and Fermi level differ, the photoemission threshold energy corresponds to the ionization potential.
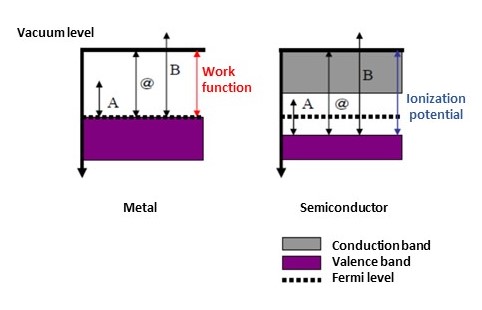
Fig.1 Relationship between band diagram and work function /
ionization potential of metal / semiconductor
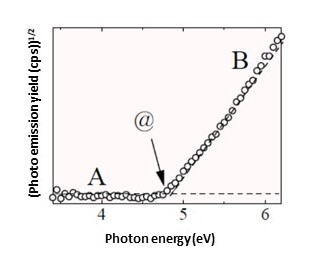
Fig.2 Measurement of work function and ionization potential